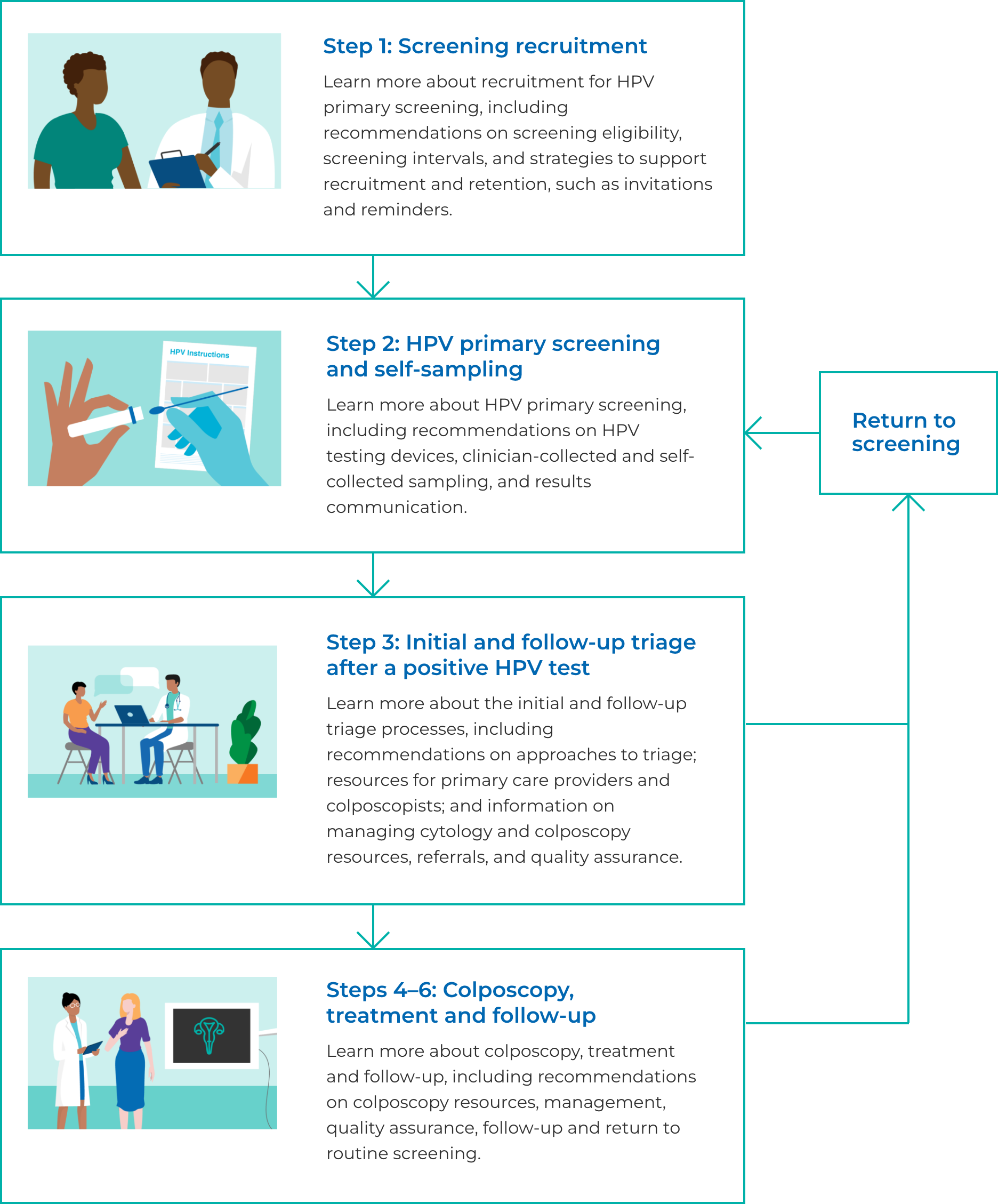
HPV screening and follow-up pathway
Centralized resources to support HPV primary screening and follow-up
Action Plan for the Elimination of Cervical Cancer
Cervical cancer is almost entirely preventable, and most cervical cancers are caused by oncogenic strains of the human papillomavirus (HPV). In response to the World Health Organization’s global call for action to eliminate cervical cancer and to support realization of the Canadian Strategy for Cancer Control, together with partners across the country, the Canadian Partnership Against Cancer (the Partnership), developed the Action Plan for the Elimination of Cervical Cancer in Canada, 2020-2030 (the Action Plan). Transitioning from Papanicolaou (Pap) screening (the current primary cervical screening test) to HPV primary screening as well as developing standardized care pathways for timely and appropriate follow-up of individuals with positive screen results are two key priorities of the Action Plan.
Cervical screening is available across all jurisdictions in Canada with organized cervical screening programs available in most jurisdictions. Many provinces and territories are working towards moving from PAP screening to the implementation of HPV primary screening within their jurisdiction. HPV infection causes almost all cervical cancers. HPV primary screening can identify risk of cervical cancer earlier and reduce the number of people who get the disease. The pan-Canadian Cervical Screening Network (PCCSN) identified an HPV primary screening and follow-up pathway as a key resource needed to support jurisdictions who are considering the implementation of HPV primary screening and follow up.
How to use this pathway
The HPV primary screening and follow-up pathway is a centralized resource developed to share evidence and real-world considerations that support provincial and territorial cancer screening partners who are implementing or considering implementing HPV primary screening and follow-up. The Pathway was developed in collaboration with pan-Canadian cervical screening partners. The Pathway contains six steps, each outlining leading national and international practices and resources (including considerations for partner engagement) that can enable equitable screening and embed cultural competency in healthcare teams.
Read recommendations for engaging with partners and local communities about HPV primary screening and follow-up.
Find out how to improve provider and participant awareness and education on HPV primary screening.
Guidelines to support follow-up after a positive HPV test
To support jurisdictions, who are planning to update their clinical guidelines, the Partnership has funded the Society of Gynecologic Oncology of Canada (GOC) to develop clinical guidelines on the management of participants with a positive HPV test and risk-based colposcopy and surveillance, with collaboration from the Society of Canadian Colposcopists (SCC). GOC guidelines are not intended to supersede local guidance.
Intended users of the pathway
The pathway was created to support provincial and territorial decision-makers and leaders responsible for the delivery of organized cervical cancer screening and follow-up care. This includes:
- provincial/territorial cancer agencies and screening programs
- provincial/territorial ministries, departments or branches responsible for cancer screening and control
- provincial/territorial laboratory service departments or branches
- research, practice and policy specialists working in cancer prevention, cancer screening, public health, cervical cancer treatment and Indigenous health, including professional organizations, community organizations and pan-Canadian health organizations
Care providers (including primary care, colposcopists and oncologists) may also find the pathway useful and important to consider as HPV primary screening is integrated in their practice.
Contributors to the pathway
The Pathway was identified as a priority by the pan-Canadian Cervical Screening Network (PCCSN). The Partnership would like to thank and acknowledge the members of the HPV Screening and Abnormal Screen Follow-up Working Group, including the patient and family advisors, and representatives in primary care, cytology, colposcopy, who provided strategic guidance and expertise to the development of this pathway.
The Partnership would also like to thank PCCSN and the Canadian Agency for Drugs and Technologies in Canada (CADTH) for input and resources shared to support the development of the pathway.
Learn more
View Canadian and international resources on HPV screening and follow-up.